Lesson 2: How Mushrooms Talk to the Immune System
Introduction
There’s something unusual about the way mushrooms affect the human body. Not quite plant, not quite animal, fungi occupy a strange and fascinating middle ground. And because of that, they know how to talk to us—on a cellular level.
Unlike plants, fungi share a surprising number of traits with humans. We have more in common with mushrooms than with apples or oak trees: similar ways of metabolizing nutrients, of sending signals from cell to cell, of defending against microscopic threats. And in some cases, we even face the same enemies. Certain bacteria, viruses, and parasites don’t just infect humans—they infect fungi too.
So it makes sense, in a way, that mushrooms might have something to teach our immune systems. That they might lend us a kind of borrowed wisdom, shaped by millions of years of shared survival.
In this lesson, we’ll explore how mushrooms communicate with our immune system—how their compounds help modulate, strengthen, and sometimes even calm down our body’s defenses. And we’ll look at how herbalists work with these fungi to support immune health, not just in moments of crisis, but as a long-term strategy for resilience.
How the Immune System Knows What to Do
The immune system is, at its core, a gatekeeper. It exists to draw the line—between friend and foe, between healthy and harmful, between what belongs and what doesn’t. That line, though, is rarely obvious.
Its job is twofold. First, it fends off intruders: bacteria, viruses, parasites—the usual suspects. Second, it keeps an eye on what’s already inside, making sure that damaged or abnormal cells don’t multiply unchecked. Think of it as both a border guard and an internal auditor.
But here’s the catch: for the immune system to do its job, it has to be perfectly calibrated. If it underreacts, infections slip through. If it overreacts, it can start targeting the body’s own tissues—what we call autoimmunity. It has to be smart, fast, adaptable, and—above all—discerning.
That discernment comes from an intricate network: white blood cells that specialize in different threats, chemical messengers that help them coordinate, and cellular receptors that allow them to ‘listen’ to what’s going on. It’s a kind of biological newsroom—decisions made moment to moment, under pressure, based on constantly shifting information.
We won’t cover every detail. But in the next section, we’ll meet the key figures in this cellular drama—especially the ones that mushrooms, intriguingly, seem to influence the most. Because if fungi have something to say to the immune system, we need to understand the language they’re speaking.
The Body’s First Responder: Innate Immunity
Imagine a break-in. The first person on the scene isn’t the detective or the forensic analyst—it’s the patrol officer. Quick to arrive. Armed with general training. Ready to act.
That’s innate immunity.
When something goes wrong in the body—say a virus sneaks in, or a cell starts to malfunction—the innate immune system is the one that shows up first. It doesn’t pause to analyze or ask questions. It just gets to work. It’s fast, broad, and decisive.
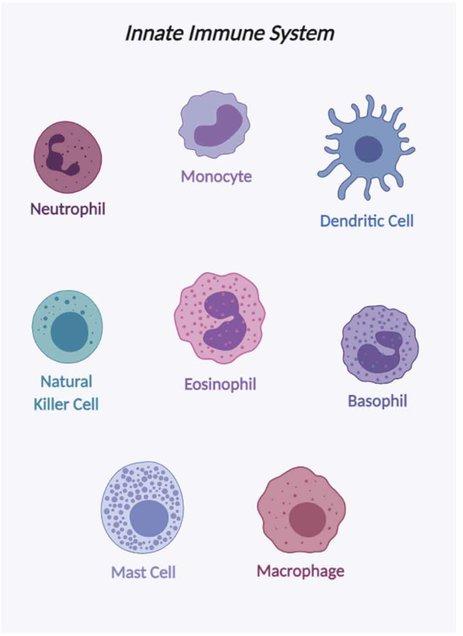
Innate immunity is made up of what scientists call “non-specific” responders. That means these immune cells aren’t on the lookout for one particular intruder. Instead, they’re trained to recognize a pattern—common red flags found on all sorts of pathogens and damaged cells.
There are macrophages and neutrophils—cells that literally gobble up invaders. There are natural killer (NK) cells, which take out infected or rogue cells without hesitation. These are the frontline defenders. They don’t hesitate. They don’t deliberate. They act.
But they’re not just brute force.
These early responders also sound the alarm. They release chemical messengers—cytokines—that call in backup. That backup is the adaptive immune system, which we’ll meet soon.
And then there’s inflammation. That redness, that swelling—it’s not just a side effect. It’s part of the strategy. Heat and pain signal that something’s wrong, while swelling brings in fluids and cells needed for repair and defense. Inflammation may be uncomfortable, but it’s often a sign that your innate immune system is doing exactly what it’s meant to do.
What’s fascinating is how this ancient part of our biology—hardwired and fast-acting—knows when to step in, and when to pass the baton. Because as effective as innate immunity is, it doesn’t work alone. It hands the case over to a more specialized team, one that takes things to the next level.
The Language of Immunity: Cytokine Conversations
If the immune system had a voice, it would speak in cytokines.
Cytokines are tiny proteins, but they carry enormous weight. They’re the messengers, the translators, the broadcasters of the immune world. When one immune cell needs to alert another—or issue instructions, or even call for reinforcements—it doesn’t shout. It sends a cytokine.
And every cell in the immune system seems to speak this language. They don’t just send messages—they listen, too. Each cell is simultaneously broadcasting and tuning in, part of a crowded, humming network of chemical communication.
Some of these signals amplify each other, like two instruments playing in harmony. Others cancel each other out, competing for airtime. It’s not just noise—it’s coordination.
This cytokine chatter is what keeps the immune response balanced. The innate system, our first responder, uses cytokines to decide how aggressively to react—and when to pass the baton to the adaptive immune team. And once that adaptive team is in play, it sends its own cytokine feedback, nudging the innate response into alignment.
You don’t need to memorize which cytokine does what. What matters is this: cytokines are how immune cells stay in sync. Without them, there would be no cohesion, no orchestration. Just chaos.
And if fungi can influence cytokine signaling—as research increasingly suggests—then they aren’t just allies. They’re fluent in the language of our immune system.
The Immune System’s Memory: How the Body Learns to Recognize Its Enemies
If innate immunity is a gut reaction, then acquired immunity is the thoughtful reply.
It doesn’t leap into action the moment trouble appears. It watches. It waits. And then it responds—with precision.
This branch of the immune system takes its time, often days instead of hours, but for good reason. It doesn’t just react to any threat—it studies it. Learns its shape. Remembers its face. And the next time that same invader shows up, the response is faster, stronger, and more focused.
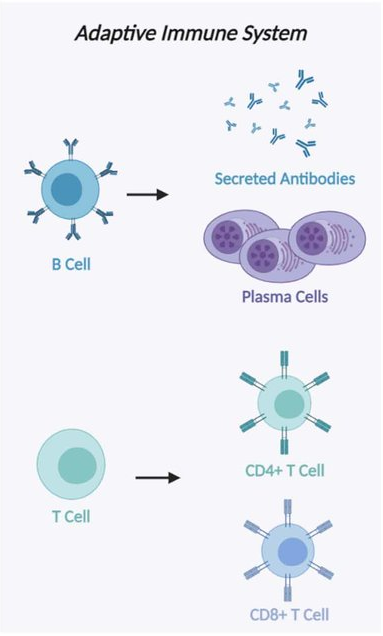
At the heart of this system are two kinds of white blood cells: B-cells and T-cells. They don’t act on instinct. They act on recognition.
B-cells are the body’s antibody factories. These antibodies are like microscopic detectives, tagging suspicious molecules—called antigens—for destruction. Once tagged, these invaders are marked for clean-up by the body’s other immune players, including the macrophages and NK cells from the innate side. It’s a cross-departmental collaboration.
T-cells are more nuanced. They don’t chase after loose antigens. They wait for a formal introduction—an antigen presented by another immune cell. Only then do they engage.
And not all T-cells fight. Some coordinate. Helper T-cells (Th cells) don’t kill invaders themselves. Instead, they orchestrate the entire response—priming B-cells, rallying cytotoxic T-cells, and ensuring that the immune system reacts with just the right amount of force.
Think of it this way: B-cells and T-cells give your immune system something it wouldn’t have otherwise—strategy. The ability not just to react, but to remember. Not just to attack, but to adapt.
It begins with a handshake. A naive helper T cell—young, uncommitted—meets an antigen complex for the first time. And from that moment, a path is chosen.
But here’s the twist: the direction that cell takes isn’t just about the antigen it encounters. It’s about the company it keeps. The chemical environment. The whispers in the cellular crowd. These signals, called cytokines, nudge the naive cell into a particular role. And that role will shape how the immune system responds—aggressively, delicately, or somewhere in between.
The two main roles are known as Th1 and Th2. They’re both necessary. They’re both powerful. But they pull the immune system in very different directions.
Th1 cells are the tactical commanders. They bolster the cytotoxic T cells—those precise assassins that target viruses and bacteria hidden inside our own cells. Th1s also recruit natural killer cells and fan the flames of innate immunity. Their focus is internal, cellular.
Th2 cells, on the other hand, turn their attention outward. They stimulate B-cells to produce antibodies, the sentries that float through blood and tissue, scanning for intruders. But there’s a downside. Th2 activity, when dominant, can overdo it. Allergies. Autoimmunity. Chronic inflammation. It’s the immune system reacting too strongly, to the wrong things.
This balance—between Th1 and Th2, between cellular and humoral response—is delicate. And this is where mushrooms, surprisingly, enter the picture. In the next section, we’ll look at how fungi, through their unique chemistry, can influence this balance. Not by shutting anything down, but by helping the system find its rhythm.
The Molecules That Speak to the Immune System
In the early days of mushroom research, one molecule stood out like a headline: beta-glucans. These long chains of sugar molecules—technically polysaccharides—were the first to show immune-boosting and even anticancer potential. Scientists were intrigued. Here was a compound, pulled from the flesh of fungi, that seemed to spark the interest of human immune cells.
But the story didn’t end there.
As research deepened, the spotlight widened. Alpha-glucans, terpenes, proteins, and a host of other small but powerful fungal metabolites began to emerge. Each one added a new layer of complexity—and possibility. Together, they form a kind of molecular chorus, influencing not just one part of the immune system, but the entire symphony: innate and adaptive alike.
Among all these compounds, fungal polysaccharides remain the best-studied and perhaps the most influential. So that’s where we’ll begin our exploration. But keep in mind: in the world of mushrooms, the magic rarely comes from a single source. It’s the interplay—the ensemble—that matters most.
The Sugar Chains That Changed Everything
We often think of sugar as simple—sweet, familiar, maybe even a little dangerous in excess. But in the world of mushrooms, sugar takes on a new identity. It becomes structure. It becomes signal. And in the case of beta-glucans, it becomes one of the most important mediators of the human immune response.
Beta-glucans are not a single molecule, but a sprawling family of polysaccharides—sugar chains with a remarkable ability to twist, branch, and bend into almost limitless shapes. That flexibility, that architectural complexity, is what gives them their power. It’s also what sets them apart from simpler beta-glucans found in grains like oats. Mushrooms build theirs differently: more branches, more linkages, more points of contact with the immune system.
These sugar chains are woven into the very walls of fungal cells. They’re everywhere. And yet, each mushroom makes them in its own unique way—lentinan from shiitake, pleuran from oyster mushrooms, grifolan from maitake. Think of them as dialects in a shared fungal language. Different accents, same core vocabulary.
The immune system listens closely. Certain cellular receptors are tuned specifically to the shapes of these branched-chain beta-glucans. The more elaborate the pattern, the more clearly the immune system hears the message. And the message is simple: pay attention.
Understanding the subtle differences between these polysaccharides matters—but for now, it’s enough to recognize them as a group. They are, collectively, one of the most powerful ways that mushrooms communicate with our immune system.
Here’s something unexpected: the beta-glucans in mushrooms are too large to enter our bloodstream. They don’t sneak past the gut wall or flood the body like a drug. They don’t need to.
Instead, they do something much subtler—and much smarter.
As they pass through the digestive tract, these fungal polysaccharides come into contact with a hidden surveillance system just beneath the surface: lymphoid tissue. This is where the immune system keeps a watchful eye. Inside that tissue are cells equipped with something called pattern recognition receptors—tiny molecular sensors that are tuned to detect danger. These receptors aren’t picky about context. To them, many mushroom compounds look suspiciously like pathogens. And so, the alarm is sounded.
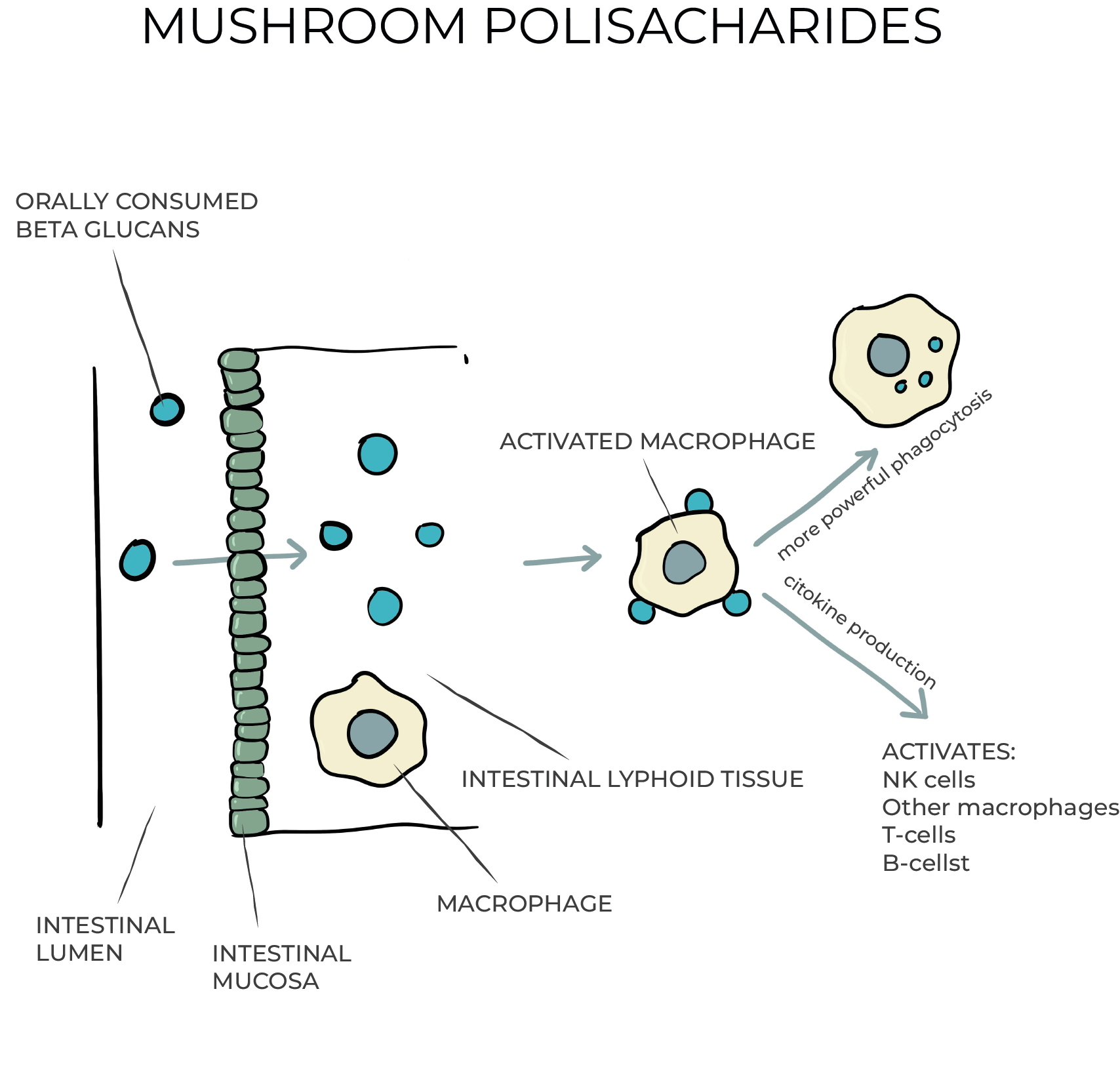
But here’s where it gets interesting. Unlike a siren that sends the immune system into a frenzy, mushrooms initiate a more nuanced response. Yes, they activate the innate immune players—macrophages, natural killer cells, dendritic cells—but not in a way that overwhelms. They nudge. They guide.
These nudges matter.
Because when dendritic cells are activated, they present antigens to T and B cells—the heart of acquired immunity. And in that moment, a decision is made: do the T-helper cells become Th1 or Th2? The direction they take determines the tone and temperament of the body’s immune response.
Fascinatingly, mushroom polysaccharides tilt that balance toward the Th1 subtype—the cellular responders. The ones that deal with viruses, with rogue cells, with the deeper housekeeping work of immunity. They don’t push the immune system into overdrive. They steer it. Gently. Strategically.
Even more surprising: this influence isn’t just about stimulation. It’s about regulation. Mushrooms help fine-tune the immune system’s internal dialogue. They increase cytokines that encourage activity, but they also dial up those that calm things down. The result isn’t immune chaos—it’s immune coherence.
And perhaps most importantly, the final outcome depends on where the body is starting from. The same mushroom may act differently depending on the person, the moment, the terrain. This is not a pharmaceutical sledgehammer. It’s a conversation. And mushrooms, it turns out, are very good listeners.
The Quiet Orchestra Inside the Mushroom
We tend to think of mushrooms as singular, simple organisms—things we sauté in butter or steep in tea. But under the microscope, they reveal themselves to be far more complex: a biochemical symphony of immune-active compounds, each playing its part in a larger orchestration. And it’s this intricate composition that makes mushrooms such remarkable allies to the immune system.
For years, polysaccharides—particularly beta-glucans—have taken center stage in the mushroom story. But they’re not the only act worth watching. In fact, there are three other equally intriguing players: terpenoids, lectins, and proteins. And while they each travel a slightly different path through the immune landscape, they seem to arrive at the same destination—enhanced innate immune activity and better-regulated inflammation.
Take lectins, for example. These are proteins that bind to carbohydrates, acting much like polysaccharides by encouraging macrophages and NK cells to become more active. They also help dendritic cells mature—a small but essential shift that allows the immune system to prepare its more complex responses. And perhaps even more intriguingly, early lab research suggests that some lectins may directly interfere with cancer cells. Whether those findings will hold true in living humans remains to be seen. But the possibility is compelling.
Then there are the terpenoids—compounds like ganoderic acid found in reishi mushrooms. These are the quiet stabilizers. They support NK cell activity and gently nudge the immune system toward a Th1-dominant state, while simultaneously tempering the kinds of inflammatory signals that can spiral out of control. Think of it as easing your foot off the gas and tapping the brakes at just the right moment.
And then there are the fungal immunomodulatory proteins—FIPs, as they’re known. These were first discovered in reishi but have since been found in other mushrooms used in Chinese medicine, like enoki, poria, and turkey tail. Like polysaccharides, they shape cytokine behavior. But FIPs may go one step further: influencing gene expression in ways that could suppress tumor growth. That’s not herbal folklore. It’s what early-stage research is beginning to suggest.
But here’s where the story deepens.
Each mushroom doesn’t just offer one of these compounds. It offers many—sometimes dozens—each interacting with different immune cell receptors, in different ways, and at different times. Most pharmaceutical research isolates a single active ingredient to study its effect. But mushrooms don’t work that way. Their power lies in their complexity—in the interplay, not the individual notes.
This is what herbalists have long called synergy: the whole acting with more intelligence and effectiveness than the sum of its parts. In mushrooms, we find a quiet orchestra, tuning the immune system not with volume, but with precision. And that, perhaps, is the most powerful kind of medicine of all.

Why Mushrooms Don’t Pick Sides
When you think of medicine, you probably imagine a substance that does one thing: kills a pathogen, lowers a fever, suppresses a symptom. It acts like a hammer. But mushrooms don’t carry hammers.
Instead, they listen.
That’s what makes them so fascinating to researchers and herbalists alike. The compounds found in fungi—particularly medicinal mushrooms—don’t just hit the immune system hard or shut it down. They modulate. Adjust. Nudge. They operate more like a thermostat than a switch, constantly checking in, recalibrating, and responding to the needs of the body. Not too much. Not too little. Just enough.
That’s why you’ll often see mushrooms described as immunomodulants—a term that tells you everything and nothing at once. In medical research, you might also come across another phrase: biological response modifiers, or BRMs. It’s a broader term, one that includes compounds that influence stress, inflammation, or even the progression of cancer. But in the world of mushrooms, BRMs are mostly about immune intelligence.
And here’s the twist: mushrooms don’t take sides. They’re just as useful when the immune system is underperforming—think chronic infections, fatigue, or immune suppression—as they are when it’s overreacting, as in the case of asthma, allergies, or autoimmune disease.
They don’t push. They reorient.
That’s the subtle brilliance of these fungal allies. Rather than acting like blunt instruments, they seem to encourage the body to use its own tools more effectively. They help macrophages clean up cellular debris. They help T-cells recognize what’s “self” and what isn’t. They calm the fire when it’s too hot—and stoke it when it’s gone out.
In other words, mushrooms don’t try to fix your immune system.
They help it remember how to fix itself.
The Quiet Strength of Immunity
If you ask someone what it means to have a “strong immune system,” you might get a vague answer—something about not catching colds or bouncing back quickly. But there’s a more precise term that immunologists use: immune competence. It doesn’t mean supercharged immunity or a body on constant high alert. It simply means your immune system knows what it’s doing. It responds when it needs to. It rests when it can.
That’s where mushrooms come in.
We’ve already explored how fungi communicate with our immune system—how their polysaccharides and proteins work more like regulators than riot police. But what does that look like in real life?
In the 1990s, researchers began studying mushrooms not just in test tubes, but in clinics. In a small group of HIV-positive individuals who couldn’t tolerate standard antiretroviral drugs, a blend of six mushroom extracts was given as the only treatment. And what happened? T-cell counts went up. Across the board. The mushrooms weren’t assisting another therapy. They were the therapy.
In another trial, this time using maitake (Grifola frondosa), results were more complex. Some patients showed little change in viral markers—but 85% of them reported they simply felt better. Fewer symptoms. More energy. A return of something hard to quantify: wellbeing.
Meanwhile, reishi (Ganoderma lucidum), one of the most studied medicinal mushrooms, was given to patients with advanced cancer. Six weeks later, their natural killer cells—the immune system’s first responders—were more active. Not superhuman. Just better equipped.
And then there’s the everyday story. The children with constant respiratory infections. In one study, they were given a beta-glucan called pleuran, found in oyster mushrooms. The result? Fewer sick days. Less worry.
What emerges from all of this isn’t a miracle cure. It’s something much more grounded: a pattern. Mushroom compounds appear to restore competence—not by overstimulating the immune system, but by helping it get back to center.
They do this slowly. Gently. Even structurally. Some mushrooms support bone marrow, the birthplace of immune cells. Others help rebuild the populations of macrophages and lymphocytes that guard the gates of health. Turkey tail, shiitake, cordyceps, maitake—they don’t rush in with sirens. They rebuild the roads.
And that’s the quiet power of immune tonics. They don’t give you something new.
They remind your body of what it already knows how to do.

When the Immune System Goes Too Far
We like to think of the immune system as a vigilant guardian. It detects threats, responds swiftly, and keeps us well. But what happens when that same system turns inward? When it mistakes friend for foe?
That’s the paradox of autoimmunity.
In conditions like rheumatoid arthritis, lupus, and Hashimoto’s thyroiditis, the immune system begins attacking the very tissues it’s meant to protect. And in conventional medicine, the answer has often been to silence the immune system altogether—with steroids, immunosuppressants, and a caution against anything that might stir the pot, including herbs like echinacea.
And yet, in East Asian medicine, mushrooms—which we’ve just learned can stimulate immunity—are often used to calm these disorders. At first glance, that doesn’t make sense. How can the same mushroom both stimulate and soothe the immune system?
Here’s where it gets interesting.
You’ll remember that helper T-cells (Th) come in different flavors. The Th2 subtype is closely tied to the production of antibodies—the very agents that drive autoimmune responses. Some mushroom extracts appear to dampen this Th2-driven cycle. They don’t shut down the immune system. They re-tune it.
A clinical trial in China put this to the test. Patients with uncontrolled rheumatoid arthritis were given a compound of reishi (Ganoderma lucidum) alongside a traditional Chinese herbal formula. The results? On paper, inflammatory markers didn’t budge. But patients felt different. They reported less pain. Doctors noticed improvements, too. In the placebo group, nothing changed.
Other small studies, this time focusing on turkey tail (Trametes versicolor) and cordyceps (Ophiocordyceps sinensis), showed promising results for lupus—again, without blunt-force suppression.
What we see here is not contradiction, but complexity. Fungi don’t operate on a single axis of “stimulate” or “suppress.” They interact. They observe. They respond.
In the world of herbalism, there’s a phrase that gets repeated often—everyone is different. Nowhere is that more apparent than in the case of autoimmune and atopic disorders. The immune system, when it misfires, doesn’t do so in the same way for everyone. It’s personal. Deeply so.
And that presents a challenge.
Mushrooms, as we’ve seen, have a remarkable ability to modulate the immune system. But even the gentlest nudge in the wrong direction can set off a storm. Some people with autoimmune conditions thrive on immune-modulating mushrooms. Others don’t. Some tolerate reishi but react poorly to cordyceps. Others find the opposite.
That’s why herbalists don’t just treat diseases. They treat people.
It’s especially important when dealing with flare-prone conditions, where the line between support and provocation is razor-thin. The answer isn’t to avoid mushrooms altogether—but to individualize the approach. To listen carefully. To observe the whole person, not just the label on their diagnosis.
The story doesn’t end with autoimmunity. Mushrooms also have something to offer for a related group of conditions: atopic disorders like allergies, eczema, and asthma. These aren’t autoimmune in the strict sense—the immune system isn’t attacking the self. But they are tied to a dominant Th2 response, the same branch of immunity responsible for hypersensitivity and allergic inflammation.
Here, mushrooms show yet another layer of complexity.
Take reishi. Long revered in East Asian medicine, this mushroom isn’t just calming—it’s chemically equipped for the task. Its triterpenoids inhibit mast cells and dial down histamine release. That means less swelling, less sneezing, fewer flare-ups.
But it’s not the only player. Clinical trials have shown that oyster mushrooms, through the beta-glucan pleuran, can lower levels of IgE and eosinophils in children with allergic tendencies. That’s not theoretical—it’s measurable. And topical pleuran creams? They’ve improved symptoms of eczema too.
Even mushrooms you’ve never heard of—enoki, aoshima, royal sun—may be quietly shifting immune balance in a helpful direction.
And this is where mushrooms truly shine: not in doing one single thing, but in their capacity to respond, adjust, and support the body’s own intelligence.
The immune system isn’t a machine. It’s a conversation. And sometimes, mushrooms are just the thing to help us change the tone.

A Balancing Act in a Cup
Sometimes, the immune system isn’t too strong or too weak. It’s both.
The same person who catches every cold that’s going around might also swell up after a mosquito bite, or sneeze relentlessly every spring. We like to think of the immune system as a dial—turn it up when you’re sick, turn it down when it’s overreacting. But it’s not a dial. It’s a network of signals, constantly adjusting. The trick isn’t amplification. It’s modulation.
This is where mushrooms shine. And this is where herbal formulas begin to speak.
Let’s look at one.
Immune-Modulating Powder Formula
This blend is designed for people who walk that tightrope between immune hyperfunction (think allergies, sensitivities, overreaction) and hypofunction (think exhaustion, recurring infections, sluggish response). And if that seems contradictory, remember: contradiction is the default setting of the human body.
Binomial Name | Common Name | Plant Part | Daily Dose | Rationale |
Astragalus membranaceus | Astragalus | Root | 3 g | Deep immune modulation; nourishing baseline support |
Ganoderma lucidum | Reishi | Fruiting body ext. | 2 g | Modulates Th1/Th2 balance; calms histamine, restores tone |
Elettaria cardamomum | Cardamom | Seed | 0.5 g | Synergist; enhances absorption and taste |
Zingiber officinale | Ginger | Rhizome | 0.5 g | Circulatory stimulant; gently moves and awakens the whole |
How to take it:
3 grams of the total powder blend, mixed into 1 fl oz of hot water, twice daily.
But let’s be clear: this isn’t a one-size-fits-all prescription. No formula ever is. What you see here is a starting point—a map, not a destination. The appropriateness of each herb depends on the person, the pattern, the terrain. Ginger may be warming to one person and too stimulating for another. Reishi may calm one system and stir another.
The real power in this formula isn’t just in the herbs. It’s in the conversation they start with the body.
The Mycelial Ally: Mushrooms in Cancer Care
For centuries, mushrooms have lingered on the fringes of healing—respected in traditional medicine, but often overlooked by modern science. That’s starting to change. Quietly, persistently, the research is stacking up. And nowhere is that shift more evident than in the realm of cancer care.
Today, mushrooms are emerging as one of the most compelling complementary tools in the support of cancer patients—not as miracle cures, but as nuanced biological allies. They’re not replacing chemotherapy or radiation. But they’re joining the conversation in a meaningful way.
What makes them so compelling?
There isn’t just one answer. There are multiple overlapping mechanisms, each with its own trail of data and promise. Some mushrooms help the immune system identify and attack cancer cells. Others appear to slow tumor growth. Still others may reduce the side effects of conventional treatments like nausea, fatigue, and immune suppression.
It’s not about a single active compound, or a one-size-fits-all model. It’s about synergy, terrain, and support. And that’s what makes mushrooms so deeply aligned with holistic care.
In the sections that follow, we’ll walk through the key mechanisms by which mushrooms may influence cancer development and progression—and how herbalists are already using this knowledge to provide grounded, respectful support to those navigating one of life’s most difficult diagnoses.
If you feel ready to dig deeper, there’s a full list of recommended readings at the end of the lesson. This is just the beginning.
The Quiet Shield: How Mushrooms May Help Prevent Cancer
Cancer begins quietly. It starts with a single error—perhaps a bit of damage to the DNA of one cell. Then, if conditions allow, that one small mutation sets off a cascade. Cells stop dying when they should. They start multiplying when they shouldn’t. And slowly, invisibly, a tumor takes shape.
What if you could interrupt that cascade before it begins?
That’s the question researchers have been asking about mushrooms. The idea isn’t new—but the science catching up to it is.
We know that oxidative stress plays a central role in cancer’s origin story. When our cells are bombarded by radiation, pollution, poor diet, or stress, free radicals are released. These unstable molecules damage DNA, sometimes permanently. And when DNA is compromised, the risk of cancer rises.
Enter mushrooms. Many species—especially their polysaccharide-rich parts—contain potent antioxidant compounds. In the lab, these compounds reduce oxidative damage to DNA, even under stressors like radiation. They act not as sledgehammers, but as shields, buffering cells against the wear and tear that can lead to malignancy.
But oxidation isn’t the only threat.
Chronic inflammation is another hidden driver. Unchecked, it creates a cellular environment where cancer can take hold and flourish. Some mushrooms—like chaga, shiitake, and oyster—don’t just dial down inflammation. They modulate it, nudging the body back toward balance, supporting immune vigilance without tipping into suppression.
Of course, this is where things get complicated. Lab results are not the same as lived experience. You can’t exactly lock thousands of people in a room for twenty years to control every variable and see who gets cancer.
But epidemiology offers quiet clues. In communities where mushrooms like enoki, shiitake, and even button mushrooms are dietary staples, the rates of certain cancers—gastric, breast—are notably lower. Correlation isn’t causation. But it’s the kind of pattern worth noticing.
Because sometimes, the most powerful interventions aren’t aggressive treatments. Sometimes, they’re slow, cumulative, and almost invisible. Like a bowl of mushroom broth shared at dinner. Or the quiet daily habit of supporting your terrain—long before anything goes wrong.
Turning the Tide: How Mushrooms Help Reawaken the Body’s First Line of Defense
Cancer, as it turns out, is not just a biological accident—it’s also an escape artist. Tumors evolve ways to dodge the immune system, silencing the very cells that are designed to find and destroy them. But what if you could flip the script? What if, instead of letting cancer hide, you could help the body remember how to see it?
This is where mushrooms come in.
Some of the most well-documented effects of medicinal mushrooms—agarikon, maitake, turkey tail, reishi, and cordyceps—relate to their uncanny ability to reawaken innate immunity. That’s the body’s non-specific, front-line defense. The one that doesn’t wait for instructions. The one that simply knows when something is wrong—and acts.
In the tumor microenvironment, though, those front-line defenders—natural killer cells, macrophages, dendritic cells—often fall silent. Cancer releases suppressive cytokines, dampening the immune response. It creates a kind of fog, and in the fog, it grows unchecked.
But mushroom compounds cut through the fog. Polysaccharides like those found in turkey tail, maitake, and reishi appear to counteract that suppression, priming immune cells to wake up, respond, and do their job—even in late-stage cancer.
Perhaps the most famous of these compounds is PSK, a polysaccharide-protein complex extracted from turkey tail mushrooms. In Japan, it’s not fringe—it’s standard. Known commercially as Krestin, PSK has been used for decades alongside chemotherapy. Not as a replacement, but as a complement. To boost immune surveillance. To improve outcomes.
And PSK isn’t the only one. PSP, its cousin, has shown similar promise. Even simple hot-water extracts of turkey tail—the kind you might prepare in your kitchen—have demonstrated immune-enhancing effects in breast cancer patients.
The point is not that mushrooms are a cure. They aren’t.
The point is that they help the body remember what it already knows how to do: defend itself. Recognize danger. Respond proportionately. And in a world where cancer’s first move is to silence the immune system, that’s a very powerful first step.

The Hidden Depths of Immunity: How Mushrooms Help Rebuild from Within
One of the most devastating side effects of cancer treatment isn’t visible on a scan or felt as pain. It happens quietly, in the marrow.
Chemotherapy—so effective at killing fast-dividing cancer cells—doesn’t discriminate. It also damages the delicate machinery of the bone marrow, where new blood cells are born. And when that production line falters, everything slows down. The immune system weakens. Anemia sets in. Fatigue deepens. The blood loses its ability to clot. Treatment becomes harder to tolerate—and sometimes, impossible to continue.
But mushrooms, in their own quiet way, may help reverse that.
Polysaccharides from maitake, shiitake, cauliflower mushroom (Sparassis crispus), chaga, and reishi appear to do something remarkable: they stimulate hematopoiesis—the generation of red and white blood cells in the bone marrow. This is what herbalists call deep immunity. Not the surface defenses that kick in during a cold, but the cellular wellspring from which all immunity flows.
In other words, these fungi don’t just support the immune system in its fight—they help rebuild it from the ground up.
And in the long journey of cancer treatment, that ability to restore what’s been depleted may be one of the most powerful contributions of all.
Slowing the Fire: How Mushrooms May Inhibit Tumor Growth
It’s one thing to boost the body’s defenses. But what if mushrooms could do something even more radical? What if they could slow the growth of cancer itself?
That’s the quiet but compelling hypothesis emerging from a growing body of research: that certain fungal compounds—especially beta-glucans—may not only support the immune system, but also interfere directly with tumor development.
How? In multiple ways. Some compounds appear to trigger apoptosis—the natural death of damaged cells. Others reduce oxidative stress, the same process that can spark genetic damage and cancer formation in the first place. Still others interrupt cellular communication, halting the cascade of signals that allow tumors to grow, spread, and invade new tissue.
Take lentinan, for example—a beta-glucan found in shiitake mushrooms (Lentinula edodes). It’s not a stand-alone chemotherapy drug. But when paired with conventional treatment, it becomes something more. Studies suggest that lentinan enhances the effectiveness of cisplatin (a widely used chemotherapeutic agent), while at the same time protecting the kidneys from cisplatin’s damaging effects. Clinical trials in lung cancer patients show a clear pattern: the combination of lentinan and chemotherapy outperforms chemotherapy alone.
A similar story plays out with maitake (Grifola frondosa). In patients with advanced-stage liver, lung, or breast cancer, maitake D-fraction—a potent polysaccharide extract—led to either tumor regression or marked symptom relief. Even more striking, when combined with whole maitake extract, the therapeutic effect was amplified, and responsiveness to chemotherapy improved.
And then there’s reishi (Ganoderma lucidum). A systematic review of its use in breast cancer found a quietly powerful statistic: patients receiving reishi were 1.25 times more likely to experience better tumor responses than those who did not.
These aren’t magic bullets. They’re not miracle cures. But they offer something just as valuable: the possibility that, through synergy and support, mushrooms may tip the scales—gently, but meaningfully—in favor of healing.
In 2010, a group of researchers in Japan asked a simple question:
What happens if you give reishi mushroom extract—every day, for a year—to people with existing colorectal tumors?
The answer was both startling and hopeful.
Patients who received the mycelial extract of Ganoderma lucidum didn’t just stop the clock on tumor progression. In many cases, they reversed it. The tumors shrank. Some disappeared entirely. Meanwhile, those who received no treatment saw their tumors continue to grow, unimpeded.
It’s the kind of clinical finding that seems almost too good to be true. And in some ways, that’s the problem.
Because the truth is: for every promising clinical trial, there are dozens—sometimes hundreds—of studies taking place in labs, not hospitals. In vitro, ex vivo, animal models. A beta-glucan might kill cancer cells in a petri dish, but that doesn’t guarantee it will do the same thing in the complex, dynamic terrain of a human body. Dose, extraction method, preparation—each of these variables can shift the outcome dramatically.
Still, the trend is undeniable. Across multiple studies, mushroom compounds consistently show the ability to slow, shrink, or suppress tumor growth. They may not act like chemotherapy—blunt-force, aggressive, often indiscriminate—but their strength may lie in something subtler. A gradual correction. A gentle rebalancing. A nudge toward the body’s own capacity to fight.
And this may be the most important insight of all: mushrooms don’t have to replace conventional medicine to matter. In fact, the best outcomes often come from integration—where herbal and conventional therapies work in tandem, side by side, each enhancing the other.
It’s not about choosing one path over another. It’s about seeing the terrain clearly—and using every tool we have to walk it wisely.
The Missing Ingredient: What Mushrooms Reveal About Healing Well
In cancer care, there’s a quiet revolution underway.
It’s no longer just about survival. It’s about how we live through the storm—and what kind of life waits on the other side.
This shift has opened the door to a different kind of medicine. One that doesn’t attack the tumor directly, but rather works in the background—restoring, supporting, buffering. Mushrooms, it turns out, are central to that story.
From shiitake to split gill, turkey tail to reishi, researchers have been testing what happens when patients undergoing chemotherapy or radiation are given mushroom extracts rich in polysaccharides. The results speak for themselves.
Less immunosuppression. Less nausea, pain, hair loss, and appetite loss. A gentler experience of an otherwise brutal process. And, perhaps most importantly, better odds of making it through—not just alive, but intact.
One study after another points to the same conclusion: patients who received mushrooms during or after conventional treatment didn’t just feel better. They did better. Their disease progression slowed. Their five-year survival rates improved.
This isn’t about choosing natural over pharmaceutical. It’s about seeing that healing isn’t only clinical—it’s experiential. And mushrooms, in their quiet, steady way, may help tip the balance from merely surviving… to living.
A Steadier Hand: Herbal Support Through Chemotherapy
Chemotherapy is not just a medical event. It’s a lived experience—one that can exhaust the body, suppress the immune system, and test the spirit.
And yet, nestled within the herbalist’s toolkit lies a quiet answer. Not a cure, not a fix—but a way to support, to buffer, and to sustain the person moving through the storm.
This formula is designed precisely for that moment.
It’s meant for use alongside conventional treatment, with the full knowledge and collaboration of an oncologist. The goal isn’t to replace, but to restore—to help the body stay upright when it wants to fall.
Chemotherapy Support Powder Formula
Botanical Name | Common Name | Plant Part | Daily Dose | Rationale |
Trametes versicolor | Turkey tail | Fruiting body extract | 4 g | Immunomodulant |
Astragalus membranaceus | Astragalus | Root | 4 g | Immunomodulant |
Ganoderma lucidum | Reishi | Fruiting body extract | 2.5 g | Immunomodulant |
Lentinula edodes | Shiitake | Fruiting body extract | 2.5 g | Immunomodulant |
Ocimum tenuiflorum | Tulsi | Aerial parts | 1 g | Adaptogen, chemoprotective |
Zingiber officinale | Ginger | Rhizome | 1 g | Antiemetic, synergist, improves digestibility |
Suggested use:
5 g mixed into 1 fl oz of hot water, 3 times daily
Every herb in this blend has a role to play. Some calm the stomach. Others protect the immune system. A few simply help the body hold on to its energy. Together, they create something greater than the sum of their parts—a quiet support structure for one of the most demanding journeys a person can face.
As always, formulas like this must be tailored with care. But in the hands of a skilled herbalist, working in concert with modern medicine, these humble plants and fungi can offer what patients so often need most: a steadier hand to hold them up.
Agarikon: The 50-Year Mushroom
In the quiet, shaded stillness of old-growth forests, something remarkable hangs from the trunks of ancient trees. Shaped like a hoof, dense as oak, and layered like the rings of time itself, Laricifomes officinalis—known as Agarikon—grows slowly, steadily, over decades. Sometimes fifty years. Sometimes more.
It prefers the company of larch, pine, and cedar—trees that have seen centuries—and it thrives where the forest is untouched, in the boreal and coastal woodlands of western British Columbia, Washington, and Oregon. Once widespread across Europe, Agarikon has retreated to these final strongholds, where it quietly continues its work.
And what is that work?

Agarikon is more than just a relic of an older world. It’s a bitter digestive tonic, a respiratory ally, and a powerful immunomodulant. Its actions stretch from antimicrobial defense to hemostatic support, making it one of the more fascinating polypores in the herbalist’s apothecary.
Each conk tells a story—season by season, layer by layer. And in those stories lies a lesson: that resilience, when rooted deeply enough, can last half a century. Or longer.
Long before antibiotics or lab-grown antivirals, there was Agarikon. In De Materia Medica—the foundational herbal text of the Western world—Dioscorides names a single fungus as a cure-all. That fungus, many now believe, was Laricifomes officinalis. Back then, it was prescribed for nearly everything: respiratory illness, menstrual pain, gut spasms, dysentery. Even tuberculosis.
Today, we’d call that a stretch. But maybe not as much as you’d think.
Agarikon has made a quiet return. Not just as folklore, but as science. Hydroethanolic extracts from its fruiting body and mycelium show antiviral action against influenza, herpes, and a host of gram-positive and gram-negative bacteria. Its bitterness—so intense it earned the nickname “quinine conk”—hints at its pharmacological potency. Herbalists now place it alongside warming digestive herbs as a restorative bitter tonic, while others turn to it for its immune-stimulating and modulating effects.
But here’s where the story takes a turn.
This isn’t a mushroom you can harvest without consequence. Agarikon lives only on the oldest trees in the oldest forests. And those forests? They’re vanishing. Which means wild Agarikon is, too. Overharvesting has already made it threatened or endangered across most of its native range.
The responsible path forward? Cultivated extracts. Ones grown in controlled conditions, away from the disappearing canopy of ancient cedar and fir. It’s a modern compromise—preserving tradition without destroying the habitat that created it.
Use wisely: High doses may upset the stomach or cause nausea. Suggested dosing is modest—200 mg to 1 g of dried decoction per day, or 1 mL of 1:5 tincture (50% alcohol), up to three times daily.
What began as a panacea is now a symbol—of endurance, yes. But also of fragility
Royal Sun: A Curious Case from the Mountains of Brazil
In the quiet highlands of Brazil, something odd caught researchers’ attention.
It wasn’t a new drug or genetic mutation. It was the elderly—walking long distances, rarely falling ill, living well into their 90s with remarkable vitality. Scientists began to investigate. Was it the air? The water? The lifestyle?
Eventually, their attention turned to a mushroom.

Locals called it Cogumelo de Deus—“the fungus of God.” To scientists, it became known as Agaricus blazei, or more recently, Royal Sun. It was part of their everyday meals—nothing exotic, just customary. But the results were anything but ordinary.
Modern research now places Royal Sun in the company of some of the most potent medicinal fungi. Its effects are as broad as they are profound: antitumor, antiviral, immunomodulating, even cholesterol- and blood sugar-lowering. It stimulates the immune system, but not recklessly—it modulates, meaning it supports balance. The same mushroom that may rally white blood cells to fight a threat can also help quiet down excessive inflammation.
Is it divine? That’s not for science to decide.
But for those seeking to support immunity, metabolism, and resilience—Royal Sun offers a compelling intersection of tradition, longevity, and emerging biomedical promise.
Sometimes, the trickiest part of working with a mushroom isn’t understanding what it does—it’s figuring out what to call it.
Royal Sun, also known as Agaricus blazei, has been caught in a scientific tug-of-war for decades. Depending on who you ask, it’s A. brasiliensis, A. subrufescens, or even a separate A. blazei entirely. Christopher Hobbs argues they’re all names for the same species. Paul Stamets leans toward renaming it. The disagreement persists. But for the herbalist or the cancer patient picking up a supplement bottle, this isn’t just academic hair-splitting. It matters. Because Royal Sun is rarely foraged—it’s purchased. And when a name is in dispute, quality control becomes critical.
What’s not in dispute is this mushroom’s promise.
Royal Sun is most famous for its potential to support cancer treatment. Lab studies show it can ramp up immune function and slow the spread of cancer cells. Its secret weapons? Immune-activating polysaccharides, yes—but also aromatase inhibitors, which may help in estrogen-sensitive cancers like certain types of breast cancer.
A clinical trial of a proprietary Royal Sun extract (AndoSan™) with patients undergoing chemotherapy for multiple myeloma found something remarkable: patients taking the extract were able to go six months longer between treatment cycles. They needed fewer IV antibiotics. They didn’t live longer—but their lives were easier.
Other studies point to reduced side effects from chemotherapy—less nausea, less fatigue, fewer mood changes. And beyond cancer, Royal Sun has a long history of use as a tonic. Today, it’s being studied for supporting heart, liver, and gut health, even in conditions like hepatitis C or Crohn’s disease. In patients with ulcerative colitis, AndoSan™ improved pain, bowel habits, energy levels, and social wellbeing.
But there’s a twist.
Like other members of its fungal family, Royal Sun contains agaratine, a hydrazine compound with potential toxicity. It sounds alarming, until you learn how fragile agaratine is. Leave the mushroom in warm water for two days and the compound disappears. Hot-water decoctions—like the traditional preparations—may be the safest and most sensible way to take this mushroom after all.
Dose: 3–5 grams of the dried fruiting body, decocted in water, three times a day. That’s what the studies used. That’s what appears to work.
Royal Sun may not be a cure, but for many people, it offers something else: a little more space, a little more ease, a little more resilience—in the middle of something very difficult. And sometimes, that’s enough.
The Secret Cord: What Makes Usnea Unique
To the untrained eye, Usnea looks like nothing more than a pale-green tangle clinging to a branch. Moss-like, wiry, maybe even forgettable. But take a closer look—split it open—and you’ll find something surprising: a fine, white, elastic thread running through its center.
That thread is a signature. A fungal core, hidden inside a body made of both fungus and algae. Usnea is a lichen, a true partnership, and this cord is what sets it apart from its lookalike cousins. It’s also the first clue that this unassuming organism is more than meets the eye.
Lichens don’t get a lot of attention in herbal medicine. They grow slowly. They resist cultivation. And yet, Usnea barbata—and its relatives—have earned a place in apothecaries for one simple reason: they work.

Antimicrobial. Immunostimulant. Vulnerary.
Usnea has a reputation for fighting infection and helping wounds heal. It’s slow-growing, yes. But its medicine runs deep—like that hidden white cord that holds it all together.
If you’ve ever pulled apart a strand of Usnea, you’ve already met its most distinctive feature: the white inner filament that makes it feel more like thread than plant. But that thread is more than structural—it hints at something deeper. A quiet complexity. Because Usnea isn’t just one thing. It’s a symbiosis. A fusion. And its medicine mirrors that.
Usnea contains two powerful classes of compounds: phenolics and polysaccharides. The former are alcohol-soluble and tend to stimulate the immune system directly. The latter, water-soluble, are more about modulation—gently nudging immune function back into balance. That means how you prepare Usnea—in alcohol or water—may shift what it does in the body.
And what it does is impressive.
Across traditions, Usnea has carved out a place in the herbal treatment of infections—especially of the respiratory and genito-urinary tracts. From bronchitis and pneumonia to herpes and HPV, this lichen shows a curious selectivity. It doesn’t act like a blunt antibiotic hammer. It acts more like a key—targeted, precise. Chinese medicine describes Usnea as clearing heat and resolving phlegm. Western herbalism calls it a styptic, an antiseptic, a wound healer.
It’s also safe. In whole form, the herb poses little risk—even if one of its isolated compounds, usnic acid, has earned some notoriety. That risk simply doesn’t translate to the herb as a whole.
Still, it’s not for everything. Because tinctures tend to amplify its immunostimulant qualities, Usnea is best thought of as an acute-response herb—something you reach for in the moment of illness, rather than a daily tonic.
And that’s fitting, really. Usnea doesn’t try to be everything. But when it acts, it acts with quiet precision. Like that thread hidden at its core.
A Balancing Act, Beneath the Surface
From the nuisance of seasonal allergies to the life-altering weight of cancer, the immune system shapes the course of our lives in ways we often overlook—until something goes wrong. And mushrooms? They have a way of stepping into that quiet imbalance and helping to make things right again.
What’s striking isn’t just that fungi can stimulate or suppress immunity. It’s that they do something more nuanced: they modulate. They listen before they act. They restore balance—not by overriding the system, but by reminding it how to work as it should.
And that, in many ways, is the hallmark of good herbalism.
What you’ve just explored is only one thread in a larger tapestry. Mushrooms don’t limit themselves to immune health. In the coming lessons, we’ll follow them into other corners of the body—into the gut, the heart, and the rhythms of daily life. Because when you look closely, you begin to see: mushrooms are not just medicine. They are collaborators in a much larger conversation about health.
Further Reading: Where Curiosity Leads Next
If this lesson has sparked your interest—and perhaps a few questions—you’re not alone. The world of medicinal mushrooms is vast, layered, and constantly evolving. Here are a few places to continue the conversation:
- Ganoderma lucidum (Reishi Mushroom) for Cancer: A Cochrane Systematic Review
A rigorous look at Reishi’s role in cancer care, from one of the most respected sources in evidence-based medicine.
Read the review - Herbs for Cancer: An Interview with Jonathan Treasure
A thoughtful audio dialogue on herbal approaches to cancer, from an experienced practitioner’s perspective.
Listen on SoundCloud - Medicinal Mushrooms—A Clinical Guide by Martin A. Powell
An essential reference for clinicians and herbalists alike, balancing science with tradition. - Medicinal Mushrooms: Ancient Remedies for Modern Ailments by Georges Halpern
A more accessible introduction that connects historical use with modern insight. - Medicinal Mushrooms in Human Clinical Studies, Part I by Solomon P. Wasser
A deep dive into clinical evidence—especially in oncology and immunology.
Read the article - The Fungal Pharmacy by Robert Rogers
Part field guide, part materia medica—this one brings fungal medicine to life. - Usnea: The Herbal Antibiotic by Christopher Hobbs
A compact, practical read on one of the most unique lichen allies in the herbal toolkit.
Let these serve not just as resources—but as invitations. Because in the world of medicinal mushrooms, every page you turn seems to open another door.
Adotey, G., Quarcoo, A., Holliday, J., Fofie, S., & Saaka, B. (2011). Effect of immunomodulating and antiviral agent of medicinal mushrooms (immune assist 24/7™) on CD4+ T-lymphocyte counts of HIV-infected patients. International Journal of Medicinal Mushrooms, 13(2), 109-113.
Biedron, R., Tangen, J.M., Maresz, K., & Hetland, G. (2012). Agaricus blazei Murill: Immunomodulatory properties and health benefits. Functional Foods in Health and Disease, 2(11), 428-447. http://www.doi.org/10.31989/ffhd.v2i11.72
Borchers, A.T., Krishnamurthy, A., Keen, C.L., Meyers, F.J., & Gershwin, M.E. (2008). The immunobiology of mushrooms. Experimental Biology and Medicine, 233(3), 259-276. https://doi.org/10.3181/0708-MR-227
Buhner, S. (1999). Herbal antibiotics: Natural alternatives for treating drug-resistant bacteria. North Adams, MA: Storey Press.
Chen, M.L., Hsieh, C.C., Chiang, B.L., & Lin, B.F. (2015). Triterpenoids and polysaccharide fractions of Ganoderma tsugae exert different effects on antiallergic activities. Evidence-Based Complementary and Alternative Medicine, 2015(754836). http://doi.org/10.1155/2015/754836
El-Enshasy, H.A., & Hatti-Kaul, R. (2013). Mushroom immunomodulators: Unique molecules with unlimited applications. Trends in Biotechnology, 31(12), 668-677. http://doi.org/10.1016/j.tibtech.2013.09.003
Firenzuoli, F., Gori, L., & Lombardo, G. (2008). The medicinal mushroom Agaricus blazei Murrill: Review of literature and pharmaco-toxicological problems. Evidence-Based Complementary and Alternative Medicine, 5(1), 3-15. http://doi.org/10.1093/ecam/nem007
Galagan, J.E., Henn, M.R., Ma, L.J., Cuomo, C.A., & Birren, B. (2005). Genomics of the fungal kingdom: Insights into eukaryotic biology. Genome Research, 15(12), 1620-1631. http://doi.org/10.1101/gr.3767105
Gao, Y., Zhou, S., Jiang, W., Huang, M., & Dai, X. (2003). Effects of Ganopoly® (A Ganoderma lucidum polysaccharide extract) on the immune functions in advanced‐stage cancer patients. Immunological Investigations, 32(3), 201-215. https://doi.org/10.1081/IMM-120022979
Girometta, C. (2019). Antimicrobial properties of Fomitopsis officinalis in the light of its bioactive metabolites: A review. Mycology, 10(1), 32-39. http://doi.org/10.1080/21501203.2018.1536680
Guggenheim, A.G., Wright, K.M., & Zwickey, H.L. (2014). Immune modulation from five major mushrooms: Application to integrative oncology. Integrative Medicine: A Clinician’s Journal, 13(1), 32-44.
Hajšlová, J., Hájková, L., Schulzová, V., Frandsen, H., Gry, J., & Andersson, H.C. (2002). Stability of agaritine – a natural toxicant of Agaricus mushrooms. Food Additives and Contaminants, 19(11), 1028-1033. http://doi.org/10.1080/02652030210157691
Hobbs, C. (1986). Medicinal mushrooms. Summertown, TN: Botanica Press.
Hobbs, C. (1990). Usnea: The herbal antibiotic and other medicinal lichens (3rd ed.). Capitola, CA: Botanica Press.
Hobbs, C. (2017). Medicinal mushrooms: A clinician’s overview. Retrieved from https://www.christopherhobbs.com/webdocs/class-handouts/various/medicinal-mushrooms-Breitenbush.pdf
Jesenak, M., Hrubisko, M., Majtan, J., Rennerova, Z., & Banovcin, P. (2014). Anti‐allergic effect of pleuran (β‐glucan from Pleurotus ostreatus) in children with recurrent respiratory tract infections. Phytotherapy Research, 28(3), 471-474. http://doi.org/10.1002/ptr.5020
Jesenak, M., Urbancek, S., Majtan, J., Banovcin, P., & Hercogova, J. (2016). β-Glucan-based cream (containing pleuran isolated from Pleurotus ostreatus) in supportive treatment of mild-to-moderate atopic dermatitis. Journal of Dermatological Treatment, 27(4), 351-354. http://doi.org/10.3109/09546634.2015.1117565
Kothari, D., Patel, S., & Kim, S. K. (2018). Anticancer and other therapeutic relevance of mushroom polysaccharides: A holistic appraisal. Biomedicine & Pharmacotherapy, 105, 377-394. http://doi.org/10.1016/j.biopha.2018.05.138
Li, E.K., Tam, L.S., Wong, C.K., Li, W.C., Lam, C.W., Wachtel‐Galor, S., … Tomlinson, B. (2007). Safety and efficacy of Ganoderma lucidum (lingzhi) and San Miao San supplementation in patients with rheumatoid arthritis: A double‐blind, randomized, placebo‐controlled pilot trial. Arthritis Care & Research, 57(7), 1143-1150. http://doi.org/10.1002/art.22994
Li, Q.-Z., Zheng, Y.-Z., & Zhou, X.-W. (2018). Fungal immunomodulatory proteins: Characteristic, potential antitumor activities and their molecular mechanisms. Drug Discovery Today, 24(1), 307-314. http://www.doi.org/10.1016/j.drudis.2018.09.014
Lull, C., Wichers, H.J., & Savelkoul, H.F. (2005). Antiinflammatory and immunomodulating properties of fungal metabolites. Mediators of Inflammation, 2005(2), 63-80. http://doi.org/10.1155/MI.2005.63
Nanba, H. (1997). Maitake D-fraction: Healing and preventive potential for cancer. Journal of Orthomolecular Medicine, 12(1), 43-49.
Oka, S., Tanaka, S., Yoshida, S., Hiyama, T., Ueno, Y, Ito, M. … Chayama, K. (2010). A water-soluble extract from culture medium of Ganoderma lucidum mycelia suppresses the development of colorectal adenomas. Hiroshima Journal of Medical Science, 59(1),1–6.
Powell, M. (2014). Medicinal mushrooms: A clinical guide. United Kingdom: Mycology Press.
Rossi, P., Difrancia, R., Quagliariello, V., Savino, E., Tralongo, P., Randazzo, C. L., & Berretta, M. (2018). B-glucans from Grifola frondosa and Ganoderma lucidum in breast cancer: An example of complementary and integrative medicine. Oncotarget, 9(37), 24837. http://doi.org/10.18632/oncotarget.24984
Stamets, P. (2002). Mycomedicinals: An informational treatise on mushrooms. Olympia, WA: MycoMedia Productions.
Takaku, T., Kimura, Y., Okuda, H. (2001). Isolation of an antitumor compound from Agaricus blazei Murill and its mechanism of action. Biochemical and Molecular Actions of Nutrients, 131(5), 1409-1413. https://doi.org/10.1093/jn/131.5.1409
Tanaka, A., Nishimura, M., Sato, Y., Sato, H., & Nishihira, J. (2016). Enhancement of the Th1-phenotype immune system by the intake of oyster mushroom (Tamogitake) extract in a double-blind, placebo-controlled study. Journal of Traditional and Complementary Medicine, 6(4), 424-430. https://doi.org/10.1016/j.jtcme.2015.11.004
Tangen, J.M., Tierens, A., Caers, J., Binsfeld, M., Olstad, O.K., Trøseid, A.M.S., … Hetland, G. (2015). Immunomodulatory effects of the Agaricus blazei Murrill-based mushroom extract AndoSan in patients with multiple myeloma undergoing high dose chemotherapy and autologous stem cell transplantation: A randomized, double blinded clinical study. BioMed Research International, 2015(718539). http://dx.doi.org/10.1155/2015/718539
Therkelsen, S.P., Hetland, G., Lyberg, T., Lygren, I., & Johnson, E. (2016). Effect of a medicinal Agaricus blazei Murill-based mushroom extract, AndoSan™, on symptoms, fatigue and quality of life in patients with ulcerative colitis in a randomized single-blinded placebo controlled study. PloS one, 11(3), e0150191. https://doi.org/10.1371/journal.pone.0150191
Thompson, I.J., Oyston, P.C., & Williamson, D.E. (2010). Potential of the β-glucans to enhance innate resistance to biological agents. Expert Review of Anti-infective Therapy, 8(3), 339-352. https://doi.org/10.1586/eri.10.10
Torkelson, C.J., Sweet, E., Martzen, M.R., Sasagawa, M., Wenner, C.A., Gay, J., … Standish, L. J. (2012). Phase 1 clinical trial of Trametes versicolor in women with breast cancer. ISRN oncology, 2012, 251632. http://doi.org/10.5402/2012/251632
Wasser, S.P. (2017). Medicinal mushrooms in human clinical studies. Part I. Anticancer, oncoimmunological, and immunomodulatory activities: A review. International Journal of Medicinal Mushrooms, 19(4), 279-317. http://doi.org/10.1615/IntJMedMushrooms.v19.i4.10
Yarnell, E., & Abascal, K. (2008). Lupus erythematosus and herbal medicine. Alternative & Complementary Therapies, 14(1), 9-12. http://doi.org/10.1089/act.2008.14105
Zhang, Y., Zhang, M., Jiang, Y., Li, X., He, Y., Zeng, P., … Zhang, L. (2018). Lentinan as an immunotherapeutic for treating lung cancer: A review of 12 years clinical studies in China. Journal of Cancer Research and Clinical Oncology, 44(11), 2177-2186. http://doi.org/10.1007/s00432-018-2718-1
